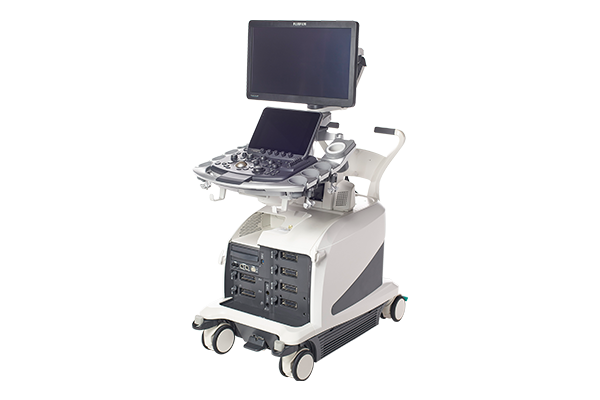
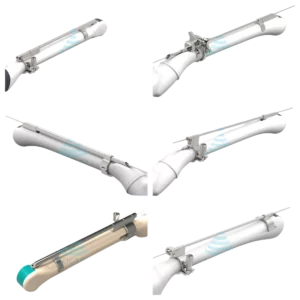
1, Understanding the Hitachi Arietta 850 Ultrasound System
The Hitachi Arietta 850 is a premium diagnostic ultrasound system engineered for advanced clinical performance across diverse medical specialties. Developed by Fujifilm Healthcare (the successor to Hitachi’s medical imaging division), this system combines cutting-edge technologies like the PureVision Plus image processing engine and proprietary elastic imaging techniques to deliver exceptional resolution and diagnostic accuracy. Designed for applications ranging from abdominal and cardiac imaging to musculoskeletal and evaluations, the Hitachi Arietta 850 caters to both hospital environments and specialized clinics seeking high-precision imaging solutions. Its modular architecture supports seamless integration with hospital networks, while its ergonomic design prioritizes operator comfort during extended use.
A standout feature of the Hitachi Arietta 850 is its adaptive beamforming technology, which dynamically optimizes sound wave transmission for clearer visualization of deep-tissue structures. This capability is particularly critical in cardiology, where accurate assessment of myocardial function relies on high-frame-rate imaging. Additionally, the system’s Real-time Virtual Sonography (RVS) module enables fusion imaging with MRI/CT scans, enhancing interventional procedure guidance. For vascular studies, the device’s high-frequency linear probes provide unparalleled detail in visualizing superficial vessels, while convex probes excel in abdominal and obstetric applications.
Clinicians favor the Hitachi Arietta 850 for its user-centric interface, which includes customizable touchscreen controls and automated measurement tools that streamline workflow efficiency. The system’s compatibility with a wide range of probes—from phased array to endocavity transducers—ensures versatility across clinical scenarios. However, maintaining probe compatibility is essential to preserve the system’s diagnostic integrity, as mismatched transducers can degrade image quality or trigger operational errors. This underscores the importance of sourcing probes specifically designed for the Hitachi Arietta 850, whether through original equipment manufacturers (OEMs) or rigorously tested third-party providers.
Beyond technical specifications, the Hitachi Arietta 850 reflects Fujifilm’s commitment to innovation through features like AI-driven artifact reduction and advanced Doppler modes for hemodynamic analysis. Its energy-efficient design and compliance with global safety standards further solidify its position as a trusted choice for healthcare institutions prioritizing both performance and sustainability. As we explore where to purchase compatible probes, understanding these technical nuances ensures informed decision-making that aligns with the system’s engineering excellence.
2, Why Choosing Compatible Probes Matters for Arietta 850
The Hitachi Arietta 850 ultrasound system delivers unparalleled diagnostic precision, but its performance hinges on using compatible probes designed to match its advanced engineering. Probes serve as the critical interface between the machine and the patient, transmitting and receiving sound waves to generate high-resolution images. For the Hitachi Arietta 850, selecting probes that align with its proprietary technologies—such as PureVision Plus image processing and adaptive beamforming—is essential to maintain optimal image clarity, depth penetration, and workflow efficiency. Mismatched or substandard transducers can compromise diagnostic accuracy, strain system components, and even void warranties, making probe compatibility a cornerstone of both clinical reliability and long-term investment protection.
One primary reason compatible probes matter lies in their technical synergy with the Hitachi Arietta 850’s architecture. Fujifilm Healthcare designs probes with specific frequency ranges, connector interfaces, and software protocols tailored to the system’s hardware. For instance, the Arietta 850’s elastic imaging modes rely on precise probe calibration to assess tissue stiffness accurately, a feature vital for liver fibrosis staging or musculoskeletal evaluations. Using non-compatible transducers may disrupt these specialized functions, leading to inconsistent data or misdiagnoses. Similarly, high-frequency linear probes optimized for vascular imaging must meet exact acoustic impedance standards to avoid artifacts in superficial structure visualization, a common pitfall with generic alternatives.
Beyond technical alignment, probe compatibility safeguards the Hitachi Arietta 850’s operational integrity. Incompatible probes often lack firmware updates or fail to integrate with the system’s AI-driven artifact reduction tools, resulting in slower processing times and subpar image quality. Over time, mismatched transducers can strain the machine’s internal circuitry, increasing the risk of hardware failures. This is particularly critical in high-volume settings like emergency departments or obstetrics clinics, where downtime directly impacts patient care and revenue. Hospitals investing in the Hitachi Arietta 850 must weigh these risks against short-term cost savings, prioritizing OEM (original equipment manufacturer) probes or third-party options rigorously tested for Arietta 850 compatibility.
Regulatory compliance is another non-negotiable factor. The Hitachi Arietta 850 adheres to stringent global standards for medical imaging devices, including FDA clearance and CE marking. Compatible probes must meet equivalent certifications to ensure safe and legal operation. Non-certified transducers may expose healthcare providers to liability risks, especially if faulty equipment contributes to adverse patient outcomes. For institutions prioritizing audit readiness and malpractice mitigation, verifying probe compliance isn’t just prudent—it’s mandatory.
Finally, cost-effectiveness plays a pivotal role. While third-party probes often carry lower price tags, their lifespan and performance variability can outweigh initial savings. Compatible probes, whether OEM or certified alternatives, typically offer better durability and support, reducing replacement frequency and service calls. For example, Fujifilm’s probes are engineered to withstand frequent sterilization processes in surgical environments, whereas incompatible models may degrade rapidly under similar conditions. By aligning probe selection with the Hitachi Arietta 850’s design specifications, facilities can balance budgetary constraints with clinical excellence.
As we delve into where to purchase compatible probes, understanding these technical, regulatory, and financial considerations ensures informed decisions that uphold the Hitachi Arietta 850’s reputation as a leader in diagnostic ultrasound technology.